Jubilant Pharmova Limited
Company Overview
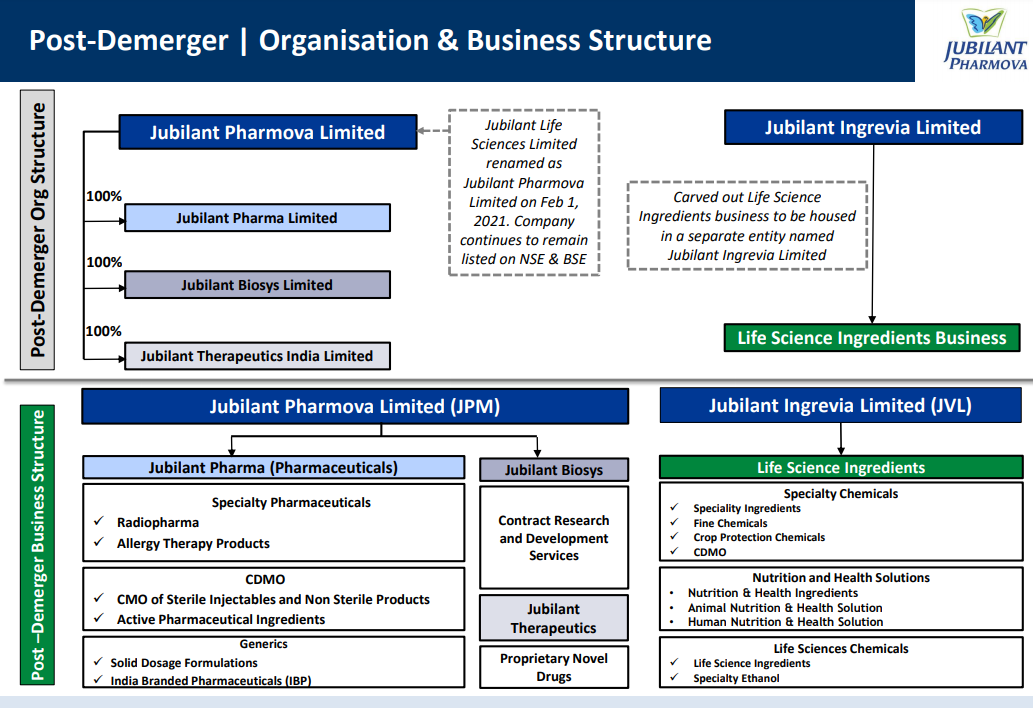
Jubilant Pharmova Limited (NSE: JUBLPHARMA) Pharmova is born out of a combination of ‘Pharma’ and ‘Nova’ (New) and Jubilant Pharmova Limited will continue to focus on offering products and services with, excellence in research and manufacturing, catering to the unmet health needs at an affordable price. 1
It is an integrated global pharmaceuticals company having three business segments Pharmaceuticals, Contract Research and Development Services and Proprietary Novel Drugs.
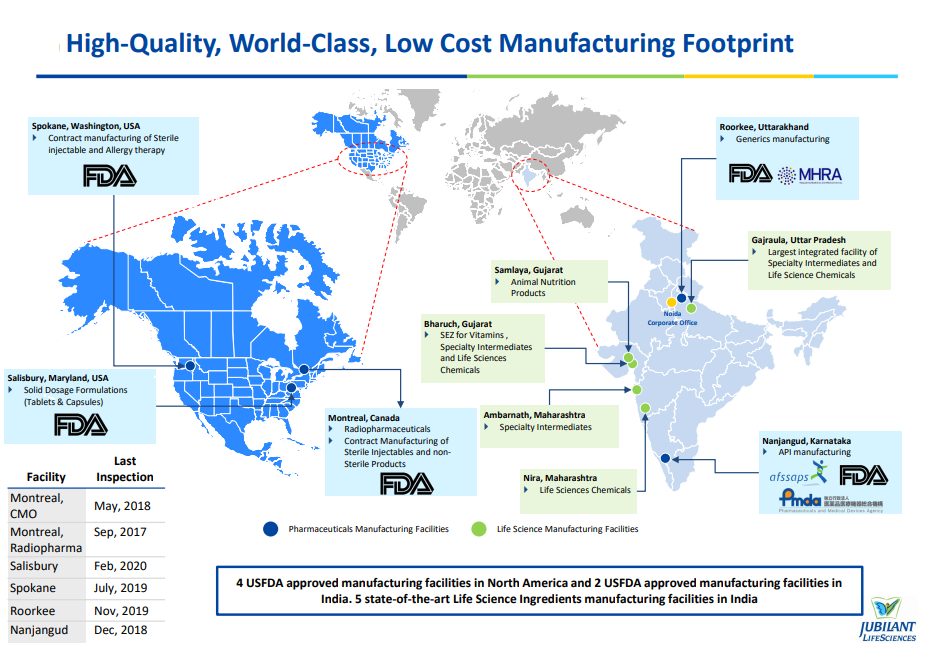
Pharmaceuticals business through Jubilant Pharma Limited is engaged in manufacturing and supply of Radiopharmaceuticals with a network of 49 Radiopharmacies in the US, Allergy Therapy Products, Contract Manufacturing of Sterile Injectables and Non-sterile products, Active Pharmaceutical Ingredients and Solid Dosage Formulations through six USFDA approved manufacturing facilities in the US, Canada and India.
Contract Research and Development Services business represented by Jubilant Biosys Limited provides innovation and collaborative research to global pharmaceutical innovators through two world-class research centers in Bengaluru and Noida in India.
Proprietary Novel Drugs business through Jubilant Therapeutics Inc. is an innovative patient-centric biopharmaceutical company developing breakthrough therapies in the area of oncology and auto-immune disorders.
Jubilant Pharmova Limited has a team of around 7,700 multicultural people across the globe and is committed to delivering value to its customers in over 100 countries. The Company is well recognized as a ‘Partner of Choice’ by leading pharmaceuticals and life sciences companies worldwide.
Business Segments
Pharmaceuticals
Jubilant Pharma Limited is a global integrated pharmaceutical company offering a wide range of products and services to its customers across geographies. The company organize its pharma business into three segments. Firstly, ‘Specialty Pharmaceuticals’ comprising Radiopharmaceuticals (including Radiopharmacies) and Allergy Therapy Products. its ‘CDMO’ segment, comprising Contract Manufacturing for Non-Oral Formulation and Active Pharmaceutical Ingredients. 2
The company supply its products and services to customers in over 85 countries. Jubilant Pharmova has four manufacturing facilities in North America and two in India, coupled with Research and Development centres in North America and India. Additionally, Jubilant Pharmova has a distribution network of more than 45 radiopharmacies in the United States.
Radiopharmaceuticals
Third largest radiopharmaceutical manufacturer in the nuclear medicine industry in the United States based on revenue
Second largest centralized commercial radiopharmacy network in the United States with a national footprint of more than 45 radiopharmacies across 22 states
Allergy Therapy Products
One of the top three players with a market share of 25% in the allergenic extract market in the United States and are currently the sole producer and supplier of venom products for the treatment of allergies in the United States
APIs
Among the leading global suppliers for several Key APIs.
Solid Dosage Formulations
One of the market leaders in the United States, based on market share of several key products
Contract R&D Services
Jubilant Biosys Ltd is a part of the Jubilant Pharmova family of companies with R&D centers in India and business offices in Asia and North America. With its global reach, Jubilant Biosys provides comprehensive drug discovery services and contract research services–from target discovery to candidate selection and with flexible business models (FFS, FTE and risk shared)–in partnership with leading worldwide healthcare companies. 3
With a clear scientific focus in oncology, metabolic disorders, central nervous system (CNS) diseases, pain and inflammation, Jubilant Biosys has rapidly emerged as a leading collaborator for pharmaceutical and biotechnology companies worldwide. In each of these therapeutic areas, Jubilant Biosys has developed a deep level of expertise in discovery informatics, computational chemistry, medicinal chemistry, structural biology, biology, in-vivo, in-vitro models and translational sciences. Combined with strong clinical development and manufacturing capabilities from other Jubilant subsidiaries, Jubilant Biosys has risen as a fully integrated contract research organization in India and headquartered at Bengaluru with end-to-end solutions.
The company's ability to be the preferred collaborator in the drug discovery domain is sustained by its commitment to comply with and continually improve its quality systems and to provide the following services:
- Contract Research
- Preclinical CRO
- Drug Discovery
- Virtual Screening
- Protein Crystallization
- Toxicology & In-vitro ADME
- Medicinal Chemistry
- Synthetic & Developmental Chemistry
- Pharmacology
Jubilant Biosys collaborates with the world’s leading pharmaceutical, biotech companies, academic institutions and research foundations.
Proprietary Novel Drugs
Jubilant Therapeutics is a patient-centric biopharmaceutical company advancing potent and selective small molecule modulators to address unmet medical needs in oncology and autoimmune diseases.
Industry Overview
As per IMF’s June 2020 World Economic Outlook update, in 2020 the global economy is expected to witness the worst recession since the Great Depression with economic output likely to contract by 4.9%. This is attributed to the damage caused by the lockdowns implemented by several large economies that resulted in disruption in supply chains, demand destruction and rapid increase in unemployment levels across several large economies. In 2020, the IMF expects the Advanced Economies to contract by 8% with the US economy likely to shrink by 8% and the Euro region by 10.2%. The Emerging markets also are expected to be severely impacted and are expected to contract by 3% in 2020 with China’s growth rate expected to decline to 1% and the Indian economy expected to contract by 4.5% in FY 2021. 4
Government and central banks across the world have resorted to unprecedented fiscal and monetary stimulus to mitigate the impact of COVID-19 and support economic activity, which is expected to lead to recovery in 2021. In 2021, IMF expects the global economy to grow by 5.4% with US and Euro region growing by 4.5% and 6% and Chinese and Indian economies growing by 8.2% and 6%, respectively. However, given the evolving pandemic situation, there is considerable uncertainty about the strength of the recovery and if the world witnesses a prolonged impact of COVID-19, then economic output and short-term outlook may further get impacted.
As per Evaluate Pharma’s annual ‘World Preview’, the global Prescription drug sales is expected to grow at 6.9% Compound Annual Growth Rate (CAGR) between 2019- 2024 to reach US$ 1.2 trillion. This growth is expected to be driven by immuno-oncology line extensions and emergence of novel technologies such as cell and gene therapy.
The Indian pharmaceutical industry is the largest exporter of quality generic drugs in the world. India currently contributes 26% by volume of generic therapeutics globally and supplies 60% of global demand for vaccines and antiretroviral (ARV) drug supplies. The Indian pharmaceutical sector’s robust set of laws and regulations, at state and federal levels, ensure highest levels of quality and safety for its drugs. India is one of the largest contributors of global biotech products and benefits from lower labor cost over other manufacturing hubs.
In March 2020, the Government of India, announced a scheme of ` 30 billion to develop three mega Bulk Drug Parks in India and a Production Linked Incentive scheme of ` 69.4 billion for promotion of domestic manufacturing of critical Key Starting Materials (KSMs)/Drug Intermediates and APIs in the country. The company believe, these are steps in the right direction and will go long way to further strengthen the domestic pharmaceuticals industry and reduce reliance on import of Key Starting Materials / Drug Intermediates from other countries. Government of India’s several initiatives announced in May 2020 aimed at promoting self-reliance of the country are also expected to boost production and 'Make in India' over the medium to long term.
According to a report by Federation of Indian Chambers of Commerce and Industry (FICCI), the Indian crop protection chemicals industry is estimated to witness a CAGR of 8.3% to US$ 8.1 billion by FY 2025, with exports growing at a higher rate of 8.6% to US$ 4.2 billion in 2025. According to an industry report, the global animal nutrition market is estimated to grow at a rate of 6.5% to reach US$ 21.4 billion by 2022.
The global crop protection chemicals market size was US$ 58.4 billion in 2019 and is anticipated to expand at a CAGR of 3.3% from 2020 to 2027. Greater emphasis on high crop output to counter food security is the key growth driver.
Financial Overview
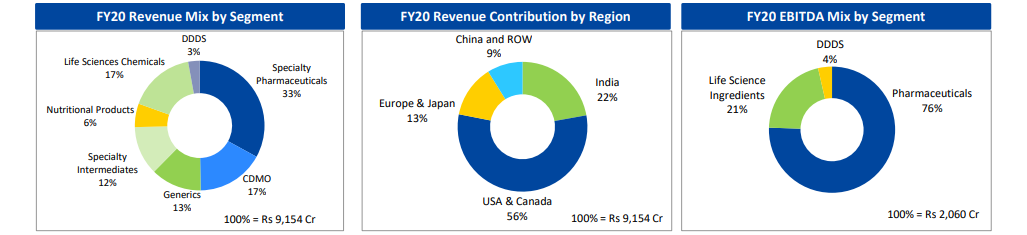
Total Revenue from Operations during the year ended March 31, 2020 stood at Rs 91,544 million as compared to Rs 91,108 million in FY 2019. Revenue from Pharmaceuticals segment grew 7% YoY at Rs 57,143 million contributing 62% to overall revenue. Revenue from Life Science Ingredients segment was at Rs 31,786 million in the year as compared to Rs 35,452 million in previous year. Revenue from Drug Discovery & Development Solutions segment stood at Rs 2,615 million in the year contributing 3% to the total revenue.
Total expenditure stood at Rs 72,073 million in FY 2020 as compared to Rs 73,718 million in the previous year. Materials cost stood at Rs 28,640 million in during the year as against Rs 32,809 million in FY 2019. Power and Fuel expense was at Rs 4,738 million in FY 2020 as compared to Rs 4,664 million in FY 2019. Employee benefit expenses stood at Rs 21,277 million during the year as compared to Rs 19,260 million in FY 2019. Other expenses were at Rs 14,652 million in FY 2020 vs. Rs 14,576 million in the previous year.
The overall EBITDA in FY 2020 grew by 12% YoY to Rs 19,945 million translating to EBITDA margin of 21.8%. The EBITDA of Pharmaceuticals segment was at Rs 15,555 million as against Rs 13,722 million in FY 2019 with margins of 27.2% as against 25.7% in FY 2019. Life Science Ingredients segment’s EBITDA was at Rs 4,310 million as compared to Rs 4,451 million in FY 2019, translating to EBITDA margin of 13.6%. Drug Discovery & Development Solutions segment EBITDA was at Rs 734 million as compared to Rs 179 million in FY 2019, translating to EBITDA margins of 28.1%.
The Profit After Tax was at Rs 8,982 million in FY 2020 up from Rs 5,745 million in FY 2019. Earnings per Share (EPS) during the year was at Rs 56.39 per equity share of Rs 1 each vs. Rs 36.86 in FY 2019.
Pharmaceuticals segment
The Pharmaceuticals segment is engaged in the manufacture, supply and distribution of Radiopharmaceuticals, Allergy Therapy Products, Contract Manufacturing (CMO) of Sterile injectables and Non-sterile products, Active Pharmaceutical Ingredients (APIs), Solid Dosage Formulations and India Branded Pharmaceuticals through six US FDA approved facilities in the US, Canada and India and contributes 62% to its Total Revenue from Operations. Revenue from this segment has improved 7% YoY to Rs 57,143 million from Rs 53,488 million last year.
Life Science Ingredients segment
Revenue contribution from the Life Science Ingredients segment to Total revenue from operations stood at 35%. During the year segment revenue was at Rs 31,786 million as against Rs 35,452 million in FY 2019.
Drug Discovery & Development Solutions (DDDS)
The Drug Discovery & Development Solutions (DDDS) segment includes its Drug Discovery Services (Jubilant Biosys Limited and Jubilant Chemsys Limited) & Proprietary Drug Discovery (Jubilant Therapeutics Inc.) businesses. Revenues in FY 2020 were at Rs 2,615 million as compared to Rs 2,168 million in the previous year, up 21% YoY.
FY21 results
Jubilant Pharmova reports net profit of Rs 214 crore in Q4; FY21 profit at Rs 836 crore. 5
Drug firm Jubilant Pharmova on Friday reported a consolidated net profit of Rs 214 crore for the fourth quarter ended March 31, 2021. The company had posted a net profit of Rs 260 crore in the January-March quarter of 2019-20.
Sales stood at Rs 1,552 crore for the fourth quarter while the same was at Rs 1,497 crore in the same period of FY20.
For the entire 2020-21 fiscal, the company reported a consolidated net profit of Rs 836 crore. It was Rs 898 crore in FY20.
Sales of the company last fiscal stood at Rs 5,964 crore, while the same was Rs 5,843 crore in FY20.
The drug firm noted that the figures for the current quarter and year were not comparable with previous periods since the Life Science Ingredients (LSI) business demerged from it with effect from February 1, 2021.
"…despite COVID-19 challenges, revenues were stable due to a diverse range of businesses. CDMO and Generics grew though the company saw impact on radiopharma and had production impact at the Nanjangud API plant," Jubilant Pharmova Chairman Shyam S Bhartia said in a regulatory filing.
Despite the pandemic-related lockdowns, the company has been able to ensure continuity in most of its manufacturing operations across all business segments while at the same time ensuring safety of employees, he added.
The drug firm said it plans to incur a capital expenditure of Rs 700-800 crore in the current fiscal on the addition of a new high speed fill and finish line with lyophilizer at Spokane site (US) and expansion of CRDS (Contract Research and Development Services) capacity. Jubilant Pharmova had a capex of Rs 276 crore in FY21.
On generics business, the company noted it plans to launch new products in the US via in-licensing and contract manufacturing.
"The company expect to launch new products from the Roorkee site once the warning letter is lifted. The company plan to enhance geographical reach in the rest of the world (RoW) markets," it added.
The company's board has recommended a dividend of Rs 5 per share of Re 1 each, aggregating to around Rs 80 crore, for the financial year ended March 31, 2021.
Business Outlook
.
Radiopharma: The company continue to build a long term pipeline of radiopharmaceuticals including Generics as well as Proprietary products being used as Diagnostic, Therapeutic, Theranostic and Devices, via in-house R&D as well as strategic partnerships with key nuclear medicine companies 6
The company expect one product launch in radiopharma in FY22. Jubilant Pharmova is further enhancing marketing and business development efforts for Ruby-Fill
Radiopharmacy: Jubilant Pharmova is executing a detailed turnaround plan of radiopharmacies to grow top line strongly with new customer wins, expand network to service newer geographies and enhance cost and procurement efficiencies. With a detailed turnaround plan of radiopharmacy, the company expect to break even in the next two-three years
Allergy: The company remains well positioned to expand volumes and growth in both venom and nonvenom extracts in the US and non-US markets over the coming years
CMO: Jubilant Pharmova has a strong visible order book of c. Rs 3,600 Crore that would be serviced over the next three years
Against Rs 500 Crore in revenues indicated earlier from the five CMO deals signed in FY21, the company realized around Rs 535 Crores. These deals are estimated to contribute further revenues of approximately Rs 200 Crore in FY22
Jubilant Pharmova is adding a high speed fill/finish line with isolator technology at Spokane site to expand capacity by 50% that will come into commercial operations by end CY24
Generics: Jubilant Pharmova has seen pricing pressure in a few products in the US. The company plan to launch new products in the US via in-licensing and contract manufacturing. The company expect to launch new products from Roorkee site once the warning letter is lifted. The company plan to enhance geographical reach in RoW markets
API: Jubilant Pharmova has a strong order book and plan to enhance volumes via new customer lock-ins
Contract Research and Development Services (CRDS): The business will continue to grow especially with upcoming commissioning of additional capacity
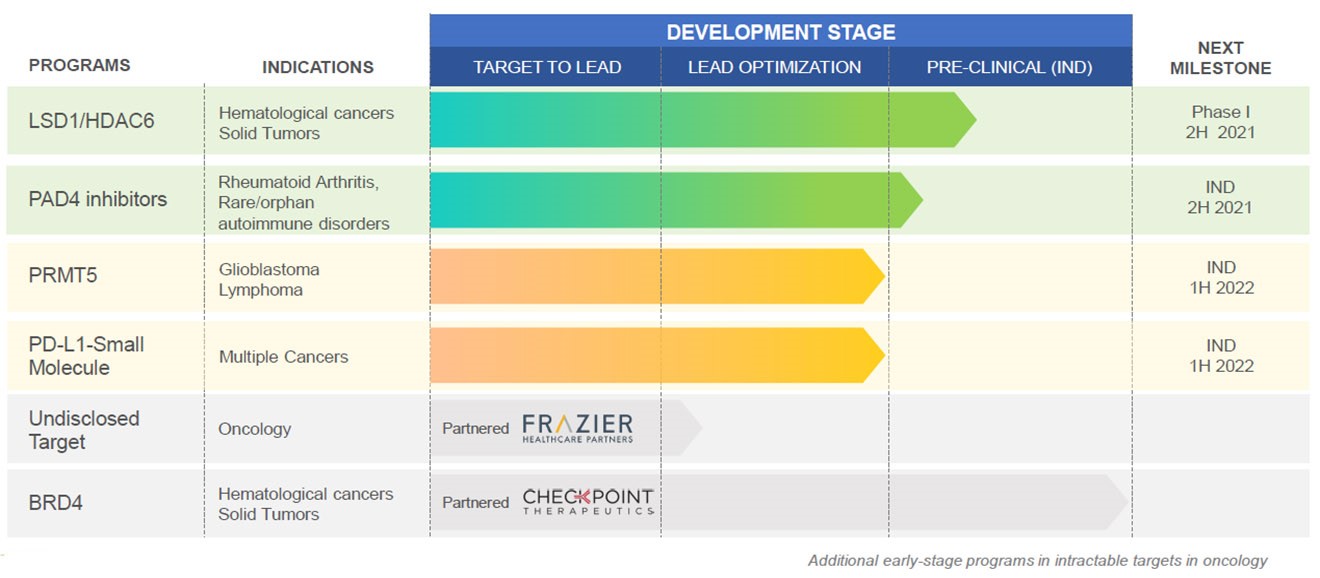
Proprietary Novel Drugs: The company plan to take one drug candidate to Phase I clinical trials in H2’FY22
Capex: The company expect to incur capex of Rs 700-800 Crore in FY22 that includes addition of a new high speed fill and finish line with lyophilizer at Spokane site and expansion of CRDS capacity
Consolidated effective tax rate: ETR of Jubilant Pharmova Limited (Continuing Operations) for FY21 is 34.1%. The company’s cash tax outflow is estimated to be at approximately 24% for the next three years. After exhaustion of the MAT credit, the Company’s effective tax rate is expected to come down to around 25% in three years’ timeframe
References
- ^ https://www.jubilantpharmova.com
- ^ https://www.jubilantpharma.com/about-us/about-jubilant-pharma
- ^ https://www.jubilantbiosys.com/about-us/company-overview/
- ^ https://www.jubilantpharmova.com/uploads/downloads/JubilantLifeSciencesAnnualReport2019-20.pdf
- ^ https://www.moneycontrol.com/news/business/earnings/jubilant-pharmova-reports-net-profit-of-rs-214-crore-in-q4-fy21-profit-at-rs-836-crore-6992441.html
- ^ https://www.jubilantpharmova.com/Uploads/files/80q1consfileQ4-FY21PressRelease.pdf




